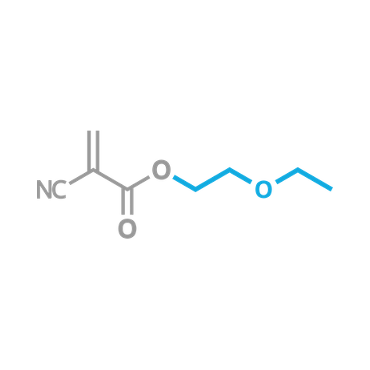
This, or the closely related 2-Methoxyethyl 2-Cyanoacrylate, is the most commonly known odorless, non-blooming liquid CA.
English
product
β-Ethoxyethyl Cyanoacrylate
categories
Monomers
Hide in products list
1