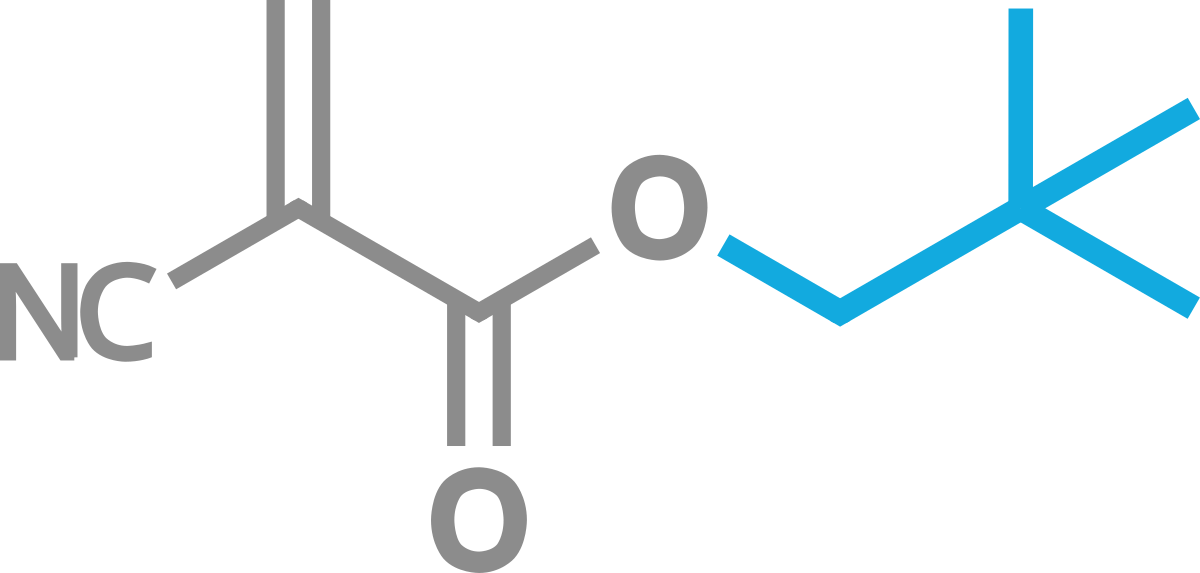
Neopentyl Cyanoacrylate
This is a non-blooming solid CA
Its synthesis and characterisation have been decribed in US 5,175,337 (Three Bond Co. Ltd.) where its use as a novel hot melt has been suggested as well as its use in liquid mixtures with other CAs. It has reported superior thermal characteristics relative to ECA. Much application detail and advantages of use for this solid CA are given in US, 6,707,107B1 (Chemence Inc.).